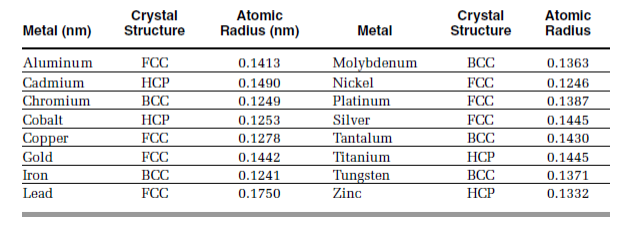
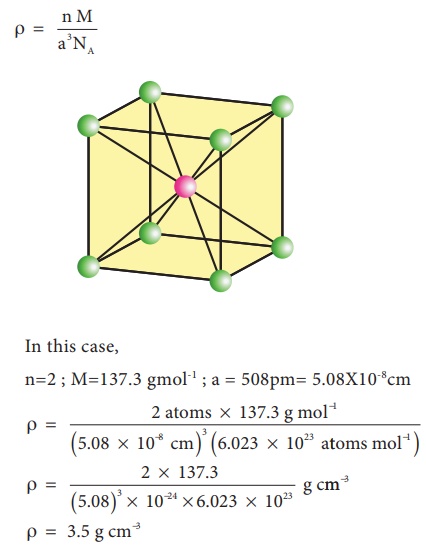
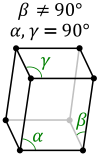
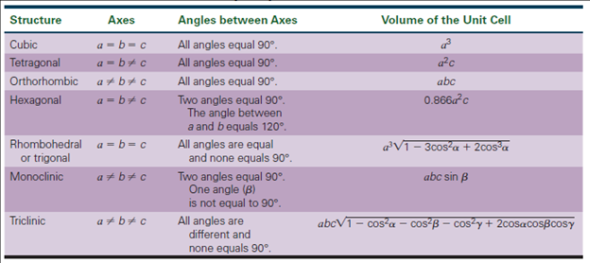
Calculate the number of unit cells in 8.1 g of aluminium if it crystallizes in a face - centred cubic (f.c.c.) structure? (Atomic mass of Al = 27 g mol^-1)
What is Atomic Packing Factor (and How to Calculate it for SC, BCC, FCC, and HCP)? – Materials Science & Engineering

SOLVED: EXAMPLE PROBLEM 3.3 Determination of HCP Unit Cell Volume (4) Calculate the volume of an HCP unit cell in lerms of its and lattice parameters (b) Now provide an expression for

Chemistry - Liquids and Solids (26 of 59) Crystal Structure: Density of the Unit Cell: Simple Cubic - YouTube